The World’s Longest-Standing, Industry-Leading Touchpoint for the Precision Medicine Ecosystem
The precision medicine field is evolving at lightning speed; from liquid biopsy-driven MRD detection and high‑plex single‑cell and multi‑omic profiling, through to breakthroughs in digital pathology, spatial analytics, and next‑generation IVD assays.
Against this backdrop the 16th World Clinical Biomarkers & Companion Diagnostics Summit returns, delivering you its largest and most comprehensive forum to date. Seize the opportunity to uncover the latest translational case studies, global regulatory shifts, and integrated commercialization strategies redefining how biomarkers power drug development, clinical decision‑making, and scalable personalized care.
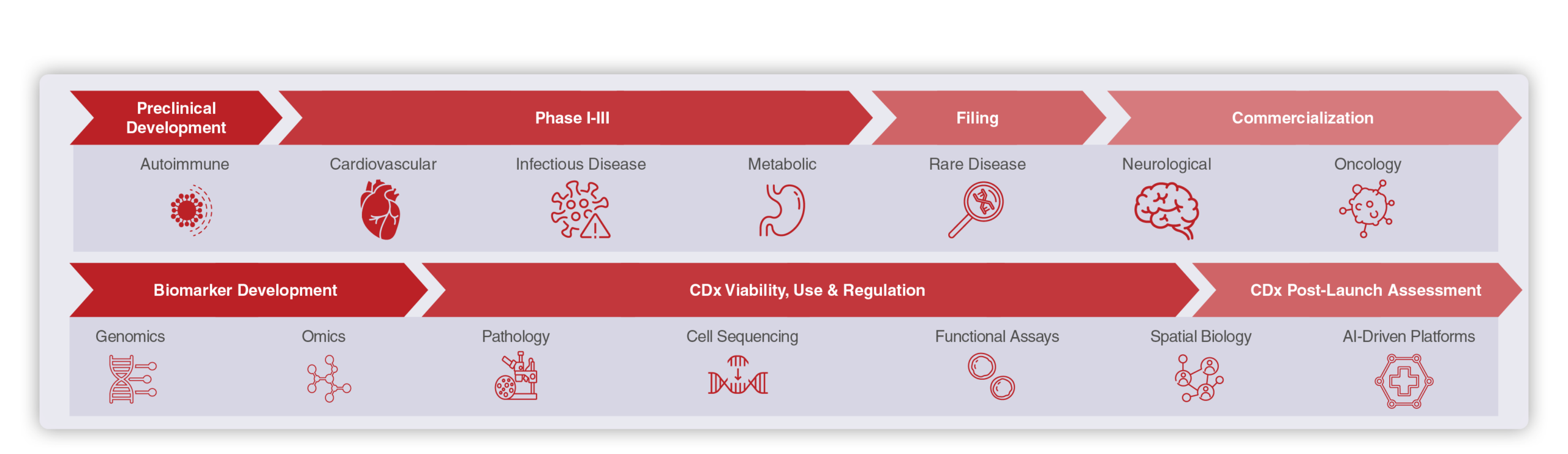
*This flowchart depicts the forum's end-to-end content, spanning the complete precision medicine co-development lifecycle
Standout Agenda Discussions Include
AI & Tech Innovators Providing Their Outlook
Accelerate precision medicine with cutting-edge AI and advanced compute harnessing omics, digital imaging, and multimodal data to drive innovation, unify complex data streams, and deliver compliant, scalable, insight-ready intelligence.
You Can Hear From:



Dynamic Precision Medicine Associations Taking to the Stage
Advancing precision medicine from bench-to-bedside demands navigating complex global regulations, reimbursement models, and healthcare systems. Hear how leading associations and charities are accelerating testing, R&D innovation, standardization, and equitable access.
You Can Hear From:





Non-Oncology Biomarkers Becoming Front & Centre
The race to elevate standards of care and enable earlier detection across neurological, autoimmune, chronic, and rare diseases is accelerating. Discover how novel biomarkers and advances in non-invasive, multimodal IVD assays are driving the next wave of precision medicine beyond oncology.
You Can Hear From:





Collaborate with 200+ World-Class Organizations
Pharma Giants
















Biotech Pioneers



















